 
The selected 2P/GSAS vaccine formulation, designated MRT5500, elicited potent nAbs as measured in neutralization assays in all three preclinical models and more importantly, protected against SARS-CoV-2-induced weight loss and lung pathology in hamsters. The lead 2P/GSAS candidate was further assessed in dose-ranging studies in mice and Cynomolgus macaques, and for efficacy in a Syrian golden hamster model. Several mRNA constructs of S-protein, including wild type, a pre-fusion stabilized mutant (2P), a furin cleavage-site mutant (GSAS) and a double mutant form (2P/GSAS), as well as others, were tested in animal models for their capacity to elicit neutralizing antibodies (nAbs). Here we describe the efforts to utilize an mRNA platform for rational design and evaluations of mRNA vaccine candidates based on the spike (S) glycoprotein of SARS-CoV-2. 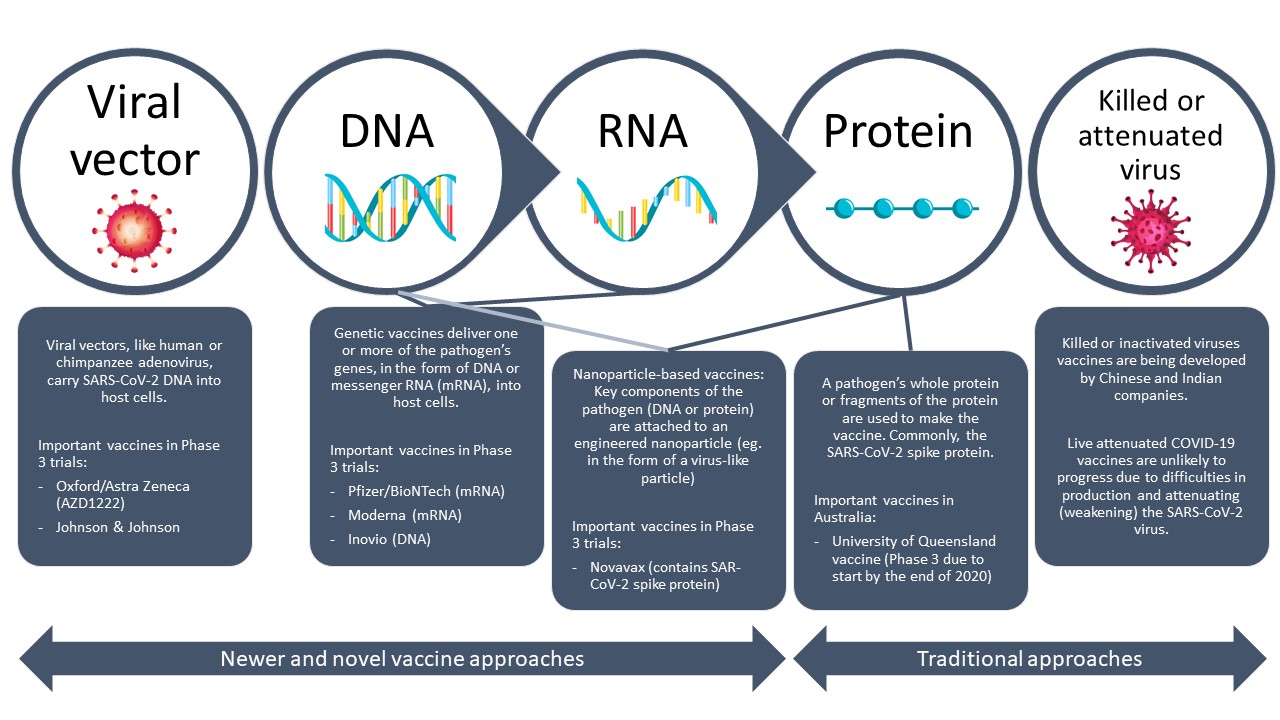
mRNA technologies offer an expeditious path alternative to traditional vaccine approaches. However, there remains a need for additional effective vaccines to meet the global demand and address the potential new viral variants. Npj Vaccines volume 6, Article number: 61 ( 2021)Įmergency use authorization of COVID vaccines has brought hope to mitigate pandemic of coronavirus disease 2019 (COVID-19). Myths and Facts about COVID-19 Vaccines.Immunogenicity and efficacy of mRNA COVID-19 vaccine MRT5500 in preclinical animal models Journal of Allergy and Clinical Immunology. Recommendations for live viral and bacterial vaccines in immunodeficient patients and their close contacts. Shearer WT, Fleisher TA, Buckley RH, et al. Duration of infectious virus shedding by sars-cov-2 omicron variant–infected vaccinees. America and Europe’s new normal: the return of vaccine-preventable diseases. Vaccination strategies in patients with solid organ transplant: Evidences and future perspectives. Vaccine safety: overview, history, and how the safety process works.Ĭenters for Disease Control and Prevention. Guidelines for the prevention and treatment of opportunistic infections in HIV-exposed and HIV-infected children.Ĭenters for Disease Control and Prevention. In: Drugs and Lactation Database (LactMed). Chickenpox (varicella) vaccine safety.Ĭenters for Disease Control and Prevention. Medical Advisory Committee of the Immune Deficiency Foundation, Shearer WT, Fleisher TA, et al. Assessing the individual risk of fecal poliovirus shedding among vaccinated and non-vaccinated subjects following national health weeks in Mexico. Scientific misconceptions and myths perpetuated in the 2017 Texas Legislative Session. doi:10.2147/RRU.S63447īaker Institute of Public Policy. 
BCG Vaccine.įuge O, Vasdev N, Allchorne P, Green JS. doi:10.1073/pnas.1400472111Ĭenters for Disease Control and Prevention. Appendix B for the Pink Book-United States Vaccine Names. General recommendation on immunization.Ĭenters for Disease Control and Prevention. Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine in adults aged 65 years and older - Advisory Committee on Immunization Practices (ACIP), 2012.Ĭenters for Disease Control and Prevention. 
Vaccine types.Ĭenters for Disease Control and Prevention. Understanding modern-day vaccines: what you need to know. Vetter V, Denizer G, Friedland LR, Krishnan J, Shapiro M. National Center for Immunization and Respiratory Diseases. Centers for Disease Control and Prevention. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
